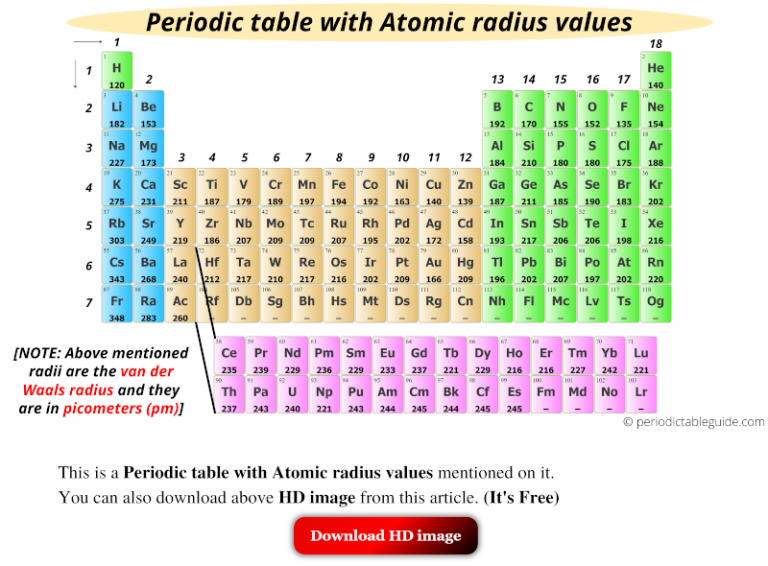
That’s why helium has a smaller atomic radius. Therefore, it shows more attraction to its electrons. This is because helium has two protons on its nucleus. Note: We must know that out of all the elements present and known so far, Helium ($He$) has the smallest atomic radius. Thus, from the given options, we can conclude that Bromine (\ ) has the smallest atomic radius as it belongs to halogens and so have covalent radius. So I can write chlorine will have smallest atomic already A and the largest. You always remember that metallic radius is always higher than that of the covalent radius. Because this is the second period element which have the list atomic radius. Moreover, the radius of alkali metals is metallic radius, whereas the radius of halogens is covalent radius.
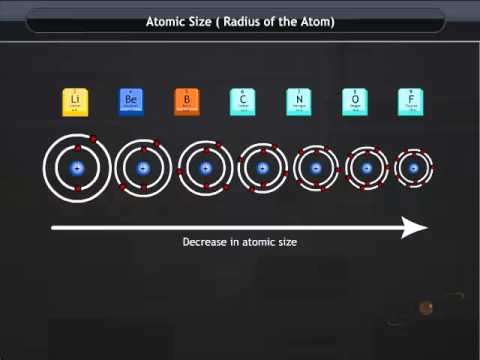
Or we can say that there is a stronger force of attraction that pulls the electrons closer to the nucleus which gives a smaller atomic radius. The concentration of more protons in the nucleus ultimately generates a higher effective nuclear charge. Atomic radius decreases as we go across a period from left to right whereas the trend of atomic radii is seen decreasing as we move up a group from bottom to top.Įlectrons are added to the same energy level, as we move across a period and at the same time, protons are being added to the nucleus. This is due to trends in the periodic table. Variations in atomic radii are observed due to the effective nuclear charge that holds the valence electrons close to the nucleus.

Group 1A of the periodic table are the alkali metals and from the given options sodium (\), potassium (\) and Lithium (\) belongs to alkali earth metals, whereas bromine (\) belongs to halogen family. The addition of electron in next level cause the atomic radii to increased. Atomic radius decreases as you move across a period.This is because Within a period, a new electron is being added to the same shell, and at the same time a new proton is added, This causes an attraction and causes the electron to pull nearer to the nucleus causing the radius to shrink and reduce in size. Atomic radii trend along group: As we move down the group atomic radii increased with increase of atomic number. The Nobel gases, Group 8 elements have the smallest atomic radIi in the periodic table. And this is because all atoms add electrons to the same shell within the periods. Explanation: The given elements are present in group one. Hint: We must know that the atomic radius decreases gradually from left to right across the period in the periodic table.
